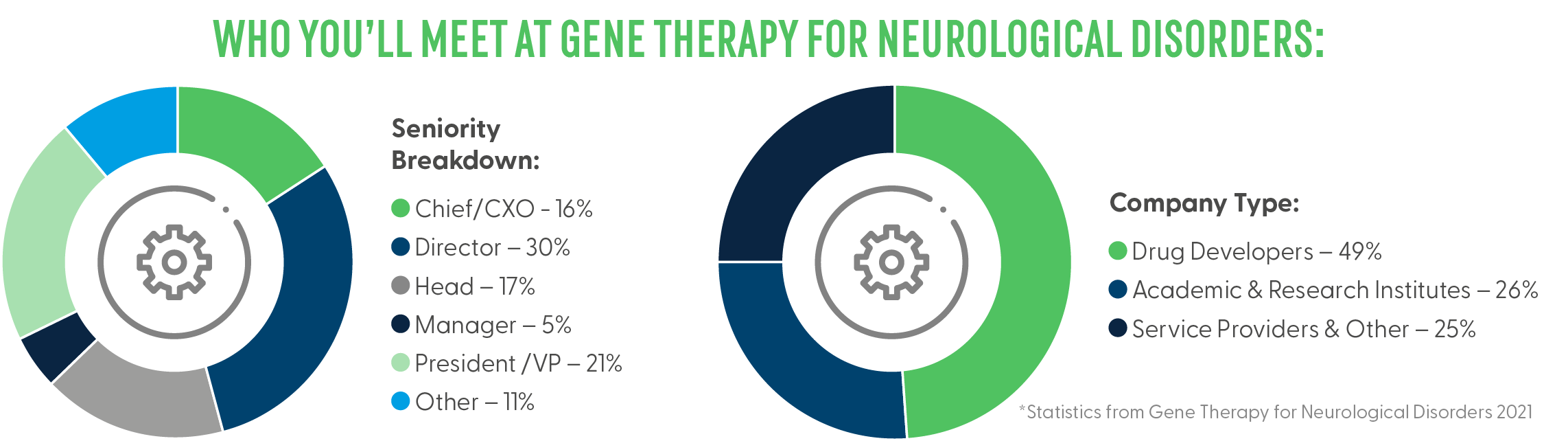
About Event
The 5th Gene Therapy for Neurological Disorders Summit
The field of neurological gene therapy development has been progressing through 2023 with a renewed sense of purpose and direction following some clinical breakthroughs, continued investment in novel vector technologies, and innovation in route of administration, with key approvals from PTC Therapeutics and bluebird bio.
To reflect the trajectory of the space this year, the 2023 post conference focus day was repurposed to the ‘Delivery Focus Day’ to allow attendees to stay on top of cutting-edge innovations in viral and non-viral vector development.
As the worlds most comprehensive industry focussed gene therapy meeting for neurological disorders, this was your opportunity to optimize your drugs to deliver safer, more efficacious gene therapies for neurological disorder patients faster.
What You Missed:
Advance your preclinical modelling systems and biodistribution studies for improved translatability when progressing to a human brain with Sanofi and Takeda
Stay on-top of cutting-edge clinical advancements and re-energize your neurological gene therapy development with PTC Therapeutics and ArgoBio
Investigate more translatable biomarkers and endpoints for CNS indications to advance clinical monitoring and improve your chances of regulatory approval with Bridge Bio and Maze Therapeutics
Identify targets and better understand disease biology in neurological gene therapy development to aid in delivery modality and drug design with Voyager Therapeutics and Lysogene
Break boundaries with novel delivery technologies and advancements in viral, non-viral and route of administration to improve distribution and efficacy with Roche, AbbVie, Capsida, and Novartis